A Ghost (Heart) of a Chance

Heir to Denton Cooley: Doris Taylor
Image: Max Burkhalter
The first thing visitors to the Texas Heart Institute’s Regenerative Medicine Research labs see is a pair of large photographs. In one, a lined hand of Denton Cooley, founder of the institute, who died last November, holds a mechanical heart much like the one that he was the first to implant into a human in 1969. In the other, Doris Taylor’s gloved hand holds a pig’s heart so stark-white, it matches her lab coat. It is a Ghost Heart, scrubbed clean of all cells, leaving only collagen, fibronectin and laminin, which provide a protein scaffold on which to build a new human heart very nearly from scratch. One day, it will cure far more patients with bum tickers than Cooley's earlier invention ever did.
Taylor and her 25-person multidisciplinary team decellularize seven or eight hearts, mostly from rats, a week, then inject the DNA-free scaffolds with stem cells. Because muscular heart cells do not divide, they cannot regenerate on their own like, say, the bladder, an organ which has been regenerated and implanted in humans. In the case of the heart, stem cells (as opposed to heart cells) adhere to the surface of the scaffold, growing into living, functioning organs inside machines known as bioreactors, which replicate the warm, oxygen-rich environment of a heart inside a mammal’s body.

A ghost heart
Image: Max Burkhalter
The goal, of course, is to build viable organs that will pump blood through an adult’s body without assistance and without the threat of rejection, since the heart will be made with the recipient’s own cells. Current testing in rats has involved implanting a second heart alongside the original, in hopes that the new organ will strengthen enough inside the body to take command.
Taylor estimates that it will be only 10 or 15 years before a functioning heart is implanted into an adult (pediatric hearts are smaller and require less muscle, so that could happen sooner)—“if we do it right,” she emphasizes. “And what I mean by that is that although it’s sexy to be first, it’s better to be best.” Taylor expects a fully functioning liver made from a recipient’s own cells will precede the heart by several years.
Along the way, Taylor and her team are regularly making discoveries in the realm of gene therapy that may prove to be just as groundbreaking as their regenerative finds. “Aging is a failure of stem cells,” she explains. Her lab is already healing damaged rat hearts with tissue patches and is secure enough in the success of such cures that she says of banking stem cells for future use. “There is no downside to it.”
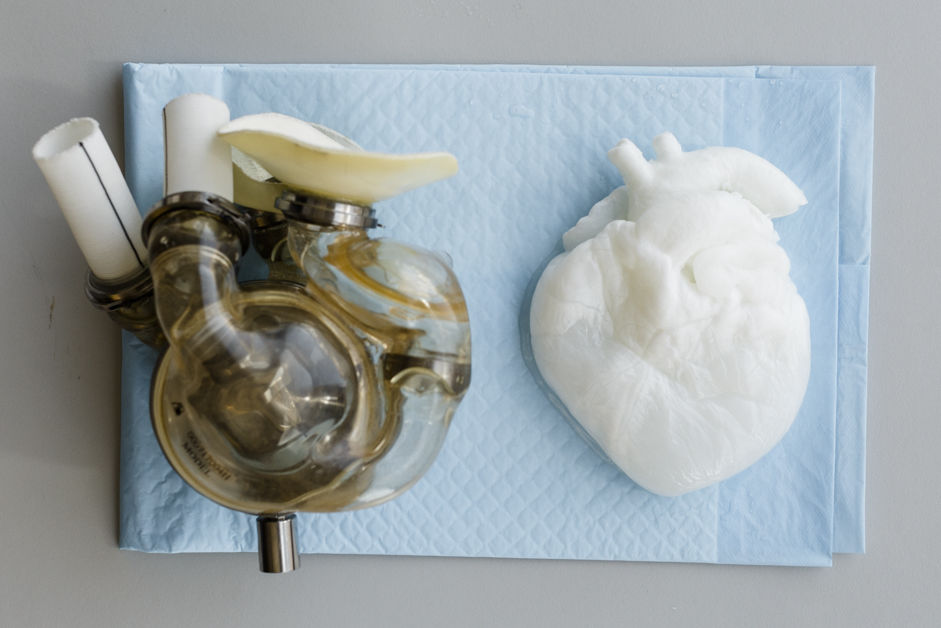
Artificial heart, left; ghost heart; right
Image: Max Burkhalter
In collecting and testing these cells, Taylor has learned that male and female ones are vastly different. Studies have revealed that female cells implanted into male rats regressed symptoms of atherosclerosis, while the inverse resulted in female rats with heightened symptoms. “Bottom line is we women are superior,” says Taylor, a mischievous glint in her eye. “There are more stem cells in the bone marrow and blood of women, and the immune system is very different than the immune system of man.”
Because women’s bodies are built for pregnancy, they’re gentler and less prone to inflammation, but also more susceptible to autoimmune diseases. Due to those findings, the lab recently received a grant to go back and chart gender differences in past cell-therapy trials. This information will help researchers learn more about how gender affects heart disease. Much of the research, Taylor explains, is made possible by last year’s 21st Century Cures Act, which, despite accusations that it would only benefit big pharma, has loosened strictures on regenerative researchers that will help speed along important discoveries in the field.
Part of the reason Taylor came to Houston four years ago was its support of work like hers. “The cool thing about Houston is philanthropy is alive and well that makes high-risk high reward possible,” she says, adding that work like hers couldn’t happen without the institute. “There are very few places in the world that are prepared to take treatments like this forward. Texas Heart Institute is one.”




