Close Encounters
The evening air was typically balmy this past May 20 when Dr. Jesse Selber arrived back in Houston. The young physician had just flown in from Philadelphia, having given a presentation to the board of directors at MD Anderson Cancer Center, which has hospitals all over the country. He works as an associate professor for the center here in Houston, in the department of plastic surgery. As he departed the plane, Selber received a phone call he’d been waiting on for well over a year. A man was dead, or very close to it—a man whose body contained everything needed to ensure another man’s survival: a healthy pancreas, kidney, scalp and skull.
An hour later, Selber was carefully examining the dying man, still on life support, at Memorial Hermann Hospital at the Texas Medical Center. “There were some issues with the scalp related to the cause of death, but separate from that, he was an otherwise good donor,” he recalls. Not only were the kidney and pancreas a match, the man’s hair and skin color closely resembled that of the patient who would soon receive a substantial portion of his head.
That patient was Jim Boysen, a 55-year-old Austinite who was first diagnosed with Type 1 diabetes at the age of 5. As in most cases of juvenile diabetes, the disease rendered his pancreas unable to produce insulin to metabolize sugars, forcing Boysen’s kidneys to compensate by working overtime. The increased stress eventually led to renal failure, 14 years ago, at which time Boysen received a dual kidney-pancreas transplant.
After that, he was placed on immunosuppressive medications, again like most patients, to prevent his body from rejecting the foreign organs. But the powerful anti-rejection drugs placed him at increased risk for developing certain types of cancer, and Boysen developed leiomyosarcoma, a rare cancer of the smooth muscle, on his scalp. Oncologists at MD Anderson were eventually able to cure the cancer, but only thanks to radiation treatment. The high doses of radiation, in turn, left him with a head wound that wouldn’t heal. Furthermore, his diabetes was causing his newly transplanted kidney and pancreas to fail. Boysen was a cancer survivor, but it was in every sense a Pyrrhic victory.
When Selber first met him in 2011, the tissue and bone surrounding the wound on Boysen’s scalp was dead “all the way down to his brain,” says the surgeon, who specializes in reconstructive procedures in cancer patients. And over the next few years, things only got worse. Boysen was caught in a Catch-22, as Selber puts it. His skull and scalp necrosis could be solved via transplants, but his kidneys and pancreas were now too weak for the patient to survive that kind of intense reconstructive surgery. And if the doctors focused instead on transplanting a new kidney and pancreas, Boysen’s worsening head wound would ensure that he wouldn’t survive that procedure either.
And then the idea came to Selber, a man whose first degree was a BA in fine arts from Brown, a non-traditional path into medicine that perhaps encouraged him to continue thinking outside the box: “I had the idea of doing a skull-scalp-kidney-pancreas transplant.” All of them, all at once, during a single, complex operative procedure. It had never been attempted before.
“It was not a conventional approach,” he chuckles. “No skin-related flap has ever been transplanted in combination with a solid organ transplant.” And that wasn’t the only obstacle to get past. While “MD Anderson is the biggest and highest-volume microvascular and reconstructive group in the world,” Selber says, “we don’t do transplant surgery. We have no expertise in that.”
And so Selber looked beyond his own hospital, to teams of doctors at other facilities in the Texas Medical Center. At first he thought he’d found a match at the University of Texas Health Science Center, where he’d already been working with a team on a facial transplant protocol. But then the transplant team there left for St. Luke’s, and ultimately the central Texas campus of Baylor Scott & White in Temple, which made a partnership impractical. Meanwhile, back at UT, a new transplant team was soon in place, but Selber found it less than accommodating. “They weren’t particularly enthusiastic,” he says.
But then, in 2013, just when he thought he’d reached a dead end, he met Dr. Osama Gaber, director of transplantation at Houston Methodist’s J.C. Walter Jr. Transplant Center. Gaber, as fate would have it, was in the audience when Selber gave a talk on behalf of LifeGift, a Houston-based nonprofit that assists patients in finding organs and tissue for transplant. He approached Selber afterward, suggesting that MD Anderson and Methodist jointly create a face transplant program. Selber liked the idea, saying he had just one caveat: “First, you’re going to have to help me with this transplant.”
Osama Gaber is an imposing yet jovial man who laughs easily and speaks in a soft Egyptian accent. “I’ve always been involved in heart-kidney, lung-liver transplants,” he says matter-of-factly. And although he admits that the idea of undertaking a quadruple transplant was “interesting,” he never expected the proposed Boysen transplant to generate such intense interest beyond the medical community. “I was like, ‘Nobody’s really interested now in transplants to the extent that we can get on the news.’ People have just gotten used to doing amazing things,” he laughs.
What intrigued Gaber and others, however, was the unusual degree of collaboration that the transplants would require. “That’s the new thing we’re doing; working together.” Gaber says he is one of a growing number of physicians who recognize that the next frontiers in medicine won’t be conquered through individual discoveries, but rather through the joint efforts of giant hospitals, medical schools and the physicians who work for them. The Texas Medical Center itself is at the forefront of this push, having recently begun work on what its website says is a “strategic planning process” to “form multi-institutional teams” in five key areas: innovation, health policy, genomics, regenerative medicine and clinical trials.
In layman’s terms, the TMC wants its institutions to take advantage of the extraordinary proximity in which they find themselves, and in so doing “position the Texas Medical Center as ‘the third coast’ for life science research, education and innovation,” says Bill McKeon, executive vice president and chief strategy and operating officer of the Center, now the largest of its kind in the world. The collective expertise of 106,000 personnel, 50,000 medical students and a physically tight-knit community of 21 hospitals and 13 other medical facilities over 1,000 acres is one of the things that makes the TMC so special, or should.
“I think the Texas Medical Center is unique,” says Gaber. “You can find everything in the Center. Everybody is at a very high level and there’s a true collaborative spirit.” Even in these days of advanced electronic communication, it seems, there’s still nothing like physical proximity. “You can go across the street and talk to your collaborator,” which is often more pleasant—not to mention productive—than any conference call or Skype session.

Jim Boysen’s multiple transplants made medical history.
Perhaps no one believes more in collaboration than Dr. Kent Osborne, who, as director of the Dan L. Duncan Cancer Center at the Baylor College of Medicine, pulls together physicians from Ben Taub Hospital, Texas Children’s Hospital, Texas Children’s Cancer and Hematology Centers and the Michael E. DeBakey Veterans Affairs Medical Center.
Founded in 2006, “the Center is unique in that it’s multidisciplinary,” Osborne says. “The days of a scientist working in a basement lab by himself are over. There’s just too much information. No one can know everything.”
In 1999, Osborne, a breast cancer specialist, was the director of medical oncology at the UT Health Science Center in San Antonio, a job he enjoyed. But then Baylor came calling, offering him not only the chance to run its new Lester and Sue Smith Breast Center, but to bring his entire team with him. “I talked to my colleagues in the breast cancer program and said, ‘It can’t hurt to hear what they have to say.’” Before long, Osborne—and almost his entire team—pulled up stakes and moved to Houston.
But while Osborne and Baylor were both thrilled with the quality of work being done at their new breast cancer center, everyone involved recognized that there was still more work to be done—and not just on breast cancer. As Osborne puts it, “we needed to have a cancer center”—one that researched everything from colon to cervical malignancies—“because cancer’s such a common illness and important area.” In 2004 he was asked to take the lead on creating just such an organization. Two years later, Houston oilman and prostate cancer survivor Dan Duncan donated $100 million to the institution that would come to bear his name on the very same day that the newly-formed Duncan Center submitted its first grant request to the National Cancer Institute.
In late May, the Duncan Center received two grants totaling $15.9 million to study liver and prostate cancers courtesy of the Cancer Prevention and Research Institute of Texas, the latest of several since CPRIT began awarding research funding to the center in January 2010. To date, CPRIT has given over $155 million to the Center, says Osborne, “because they recognize the ever-increasing importance of collaboration.” Thanks to that kind of funding, he says, “we’ve been able to help others do their research by setting up core resources that the individual would never be able to afford in their own laboratory. Nobody would be able to afford a $500,000 microscope in the lab, but the Cancer Center can because we have the grants to do it.”
It’s one thing for affiliated hospitals to come together and serve the community, as the Duncan Center has done. It’s another for rivals to do it—especially when those hospitals are as massive as MD Anderson and Memorial Hermann. But that’s exactly what these two behemoths have done with their new partnership designed to bring breast cancer care and diagnostic services to neighborhoods across Houston.
Last August, the two heavy-hitters announced a joint venture called MD Anderson Breast Care with Memorial Hermann, with plans to expand to 12 sites across Houston by the end of this year. Within just the first three months, the clinics have performed 18,000 mammograms and 30,000 procedures, diagnosing 100 patients with various cancers who “were able to get the right care at the right location,” says Amy Hay, vice president of business development at MD Anderson. And the clinics—despite their name—aren’t just focusing on breast cancer. They’re also providing screenings for gastrointestinal and lung malignancies. “Those are all cancers where if we can diagnose patients earlier,” Hay says, “we can have a much greater impact on the care they receive and their life expectancy.”
As with the Jim Boysen transplant and the Duncan Center, the MD Anderson-Memorial Hermann partnership was rooted in the understanding that each hospital had its limitations.
MD Anderson “has historically been Med Center–centric” and didn’t reach out to the Houston area at large, says Jim Brown, vice president and CEO of ambulatory services at Memorial Hermann. His own hospital, meanwhile, possessed a large community presence but its cancer screening clinics weren’t running as smoothly as they could. “Memorial Hermann historically was serviced by multiple radiology groups, and our goal was to become more consistent in how we provide that service across the city.” And so, in 2013, the hospital approached MD Anderson to propose a mutually beneficial arrangement.
“We are the global leader in cancer care,” says MD Anderson’s Hay, “but we’re in a unique position because we focus solely on cancer. Collaborating with other organizations allows us to have a much greater impact.” By going into extant Memorial Hermann clinics and integrating its own staff of radiologists, MD Anderson has found that they are able to have what Hay calls “an immediate impact.” The infrastructure was there. MD Anderson just needed to step in and steer the ship.
By all accounts, the partnership has been a success. Still, hurdles remain. The term “patient-centered” comes up a lot in conversations with both Brown and Hay, who would like to eventually see treatment plans streamlined to the point that, as Hay puts it, recently diagnosed patients “have clarity about the next step in their journey by the time they leave that day.”

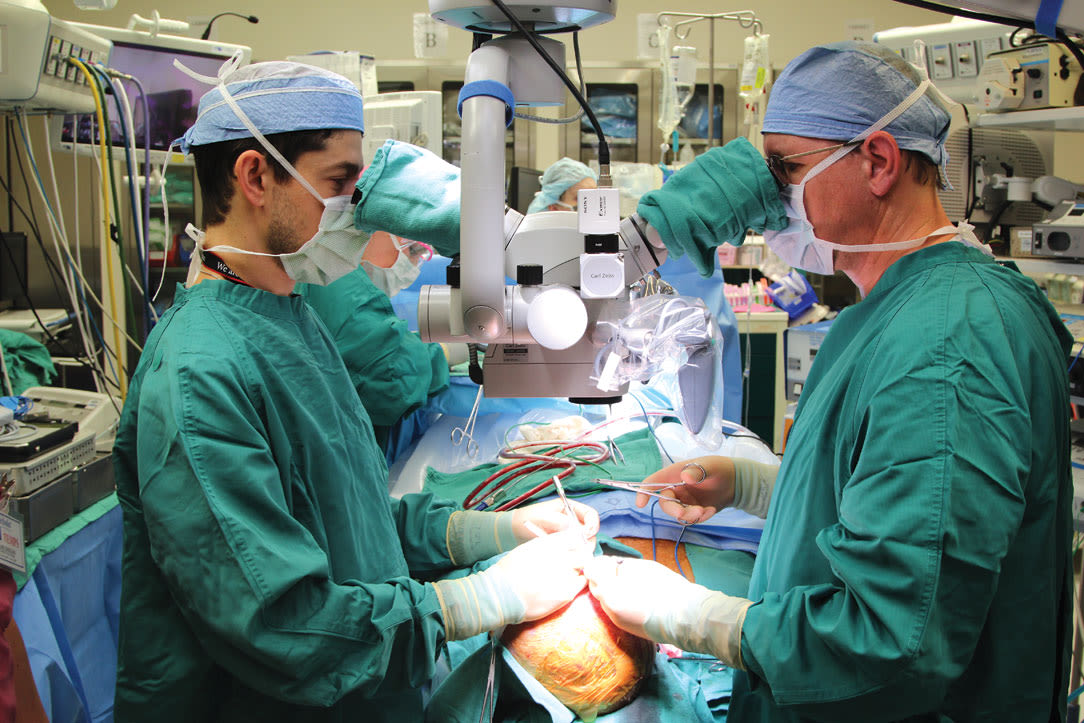
Dr. Jesse Selber and Dr. Michael Klebuc perform one of Boysen’s many surgeries.
Image: Courtesy Houston Methodist
While not denying that patients are at the center of the two hospitals’ collaboration, MD Anderson’s Selber says that the institutions themselves benefit greatly too, which can be both good and bad. Enormous untapped potential in the Texas Medical Center goes to waste, he says, when institutions, hospitals and medical schools create partnerships that benefit themselves first and foremost.
“There’s always politics that plague collaborations between institutions,” he says. “The administrators like [collaboration] to the extent that it creates publicity for them, but they want that publicity to remain in their institutions. They want to monetize everything.”
And though the Texas Medical Center has made great strides, it’s “not collaborative, particularly,” he continues. Much-touted initiatives, like the BioScience Research Collaborative, created to encourage further cooperation between Rice University and TMC physicians, for instance, have done much, funding 10 floors of research laboratories and expensive equipment like mass spectrophotometers and transmission electron microscopes costing upwards of $100,000 each.
But encouraging fuller, more fruitful collaborations depends on more than state-of-the-art facilities, says Selber. It takes people—lots of them. “We need to start developing multi-disciplinary consortiums around major medical problems where groups can discuss clinical solutions.” By way of example, he mentions the Jim Boysen case. “This is a medical center that has a rich tradition of transplant innovation. [Boysen’s complex transplant procedure] is in the tradition of that transplant innovation, and I think there’s more to come for that.”
In the 18 months following his first meeting with Selber, Gaber spent many days walking back and forth across the street, so to speak, as the two strategized a plan of attack, recruiting microsurgeons and nurses and administrators on both sides; practicing the simultaneous harvesting of kidney, pancreas, skull and scalp on research cadavers; holding near-endless meetings between institutions; and making flow charts of the exact steps they’d all have to take, at a moment’s notice, when and if a donor for Boysen was found.
As time went on, Gaber noticed his Methodist team becoming more and more devoted to the collaboration. “To use people from outside the discipline and add them to the big team and get everyone to see the value of planning … I couldn’t have spent enough money on courses and teachers to get people to believe in that.”
And then, on May 22, the entire plan sprang suddenly and swiftly into action. Not long after completing the organ harvest from the donor, Selber and a team of surgeons, nurses and other clinicians from the two hospitals—including Methodist’s Dr. Michael Klebuc, a specialist in microsurgery and facial paralysis surgery—spent several painstaking hours in the operating room removing Boysen’s necrotic skull and scalp tissue and stitching him back together again with new bone and tissue.
“It was pretty exciting, I’ve got to admit,” says Klebuc, who has a youthful voice that belies over two decades of experience. “The logistics involved in this are pretty amazing. We had a team of microsurgical nurses available on-call 24 hours a day, seven days a week…for over a year waiting for this to happen.” And when it finally did, seeing the practitioners spring into action—a team of a dozen surgeons and over 40 other healthcare professionals supporting the surgical work of Klebuc, Selber and others—was like watching the inner workings of a precision Swiss timepiece.
“What people don’t understand about these transplants is that you can’t just take a large piece of bone and tissue,” says Klebuc. “We had to connect little blood vessels about 1/16th of an inch under a microscope. And then, one of the most spectacular, magical moments you could ever see: this tissue that was grey and cold and lifeless…you could see it become pink and robust.” He sighs at the memory. “Everybody was smiling and there was this great sense of satisfaction.”
The procedure wasn’t over quite yet, however. Boysen still needed a pancreas and kidney, organs that couldn’t be transplanted until the plastic surgeons finished their work on the patient's head. From a few feet away, Dr. Osama Gaber and his team watched and waited eagerly for their turn, all the while fascinated by the history they were witnessing on the other side of the glass. “When you take a piece of bone,” says Gaber, “it’s shaped by the brain underneath it, so each one of us would have a different shape. Watching them in the operating room, I was just like, ‘How in the world are they going to get this to fit there?’”
Not long afterward, Gaber was making history of his own, having created an ingenious solution to the problem of getting blood to flow into the new pancreas and kidney post-transplant. The way to establish a good connection between Boysen’s blood vessels and those of the donor organs, Gaber believed, was to use the blood vessels of the previous donor, which were already hooked up and functioning. “We just took the [kidney] tissue and skeletonized the blood vessels and used them to put the new kidney in.” In effect, the new kidney was made to function by attaching it to the blood vessels of the previous donated kidney; ditto the transplanted pancreas. To the best of Gaber’s knowledge, it was a hitherto untried method of facilitating a kidney transplant, a process that may never have occurred to him, he says, if not for the unusual circumstances.
The list of medical discoveries made during the 24 hours of surgeries doesn’t stop there, Selber says. The surgeons also observed something they hadn't seen before about the inner surface of the skull—namely that it bleeds, and that blood is supplied by vessels near the ear. “We discovered things about the vascular supply of the skull that we would never know under ordinary circumstances.” Additional mysteries remain to be solved, such as how and why Boysen’s newly transplanted scalp was already sweating a few hours after surgery. “The sweat glands are controlled by nerves and all the nerves [were] cut,” marvels Selber.
In other words, Jim Boysen is a living piece of medical history. More importantly, Selber says, more than a month after the procedure, Boysen was doing better than ever. “His scalp looks like it was made for his head. His organs are functioning pretty well. He’s a remarkable individual.” And while it’s the dozens of doctors from two hospitals that deserve much of the credit for the transplants’ success, he says, nothing would have been possible if Boysen himself hadn’t proved to be such an extraordinary collaborator. “We had a lot of trouble along the way,” says the surgeon, reflecting on the 18 grueling months that led up to the evening of May 20.
“He kept me going, to be honest. The whole thing happened because of the kind of person he is. He could have said at any point, ‘This is getting too risky for me.’ But he didn’t. He stuck with it. And that’s what allowed us to succeed.”













































